Holey Carbon Allotropes
materials and coatings
Holey Carbon Allotropes (LAR-TOPS-165)
Innovative manufacturing methods for bulk preparation of holey graphene and holey carbon nanotubes
Overview
Researchers at NASA have developed new methods to manufacture carbon materials (e.g., nanotubes, graphene) with holes through the graphitic surface of the particles. The methods generate materials with increased accessible surface area, increased functional groups at damage sites, and improved through-surface molecular transport properties. The materials generated using these techniques are anticipated to be applicable to a variety of industries, especially energy storage (e.g. super-capacitors and batteries) and separation membranes (e.g. for gas, ions, organics, proteins, etc.).
The Technology
This invention is for scalable methods that allows preparation of bulk quantities of holey nanocarbons with holes ranging from a few to over 100 nm in diameter. The first method uses metal particles as a catalyst (silver, copper, e.g.) and offers a wider range of hole diameter. The second method is free of catalysts altogether and offers more rapid processing in a single step with minimal product work-up requirements and does not require solvents, catalysts, flammable gases, additional chemical agents, or electrolysis. The process requires only commercially available materials and standard laboratory equipment; and, it is scalable. Properties that can be controlled include: surface area, pore volume, mechanical properties, electrical conductivity, and thermal conductivity.
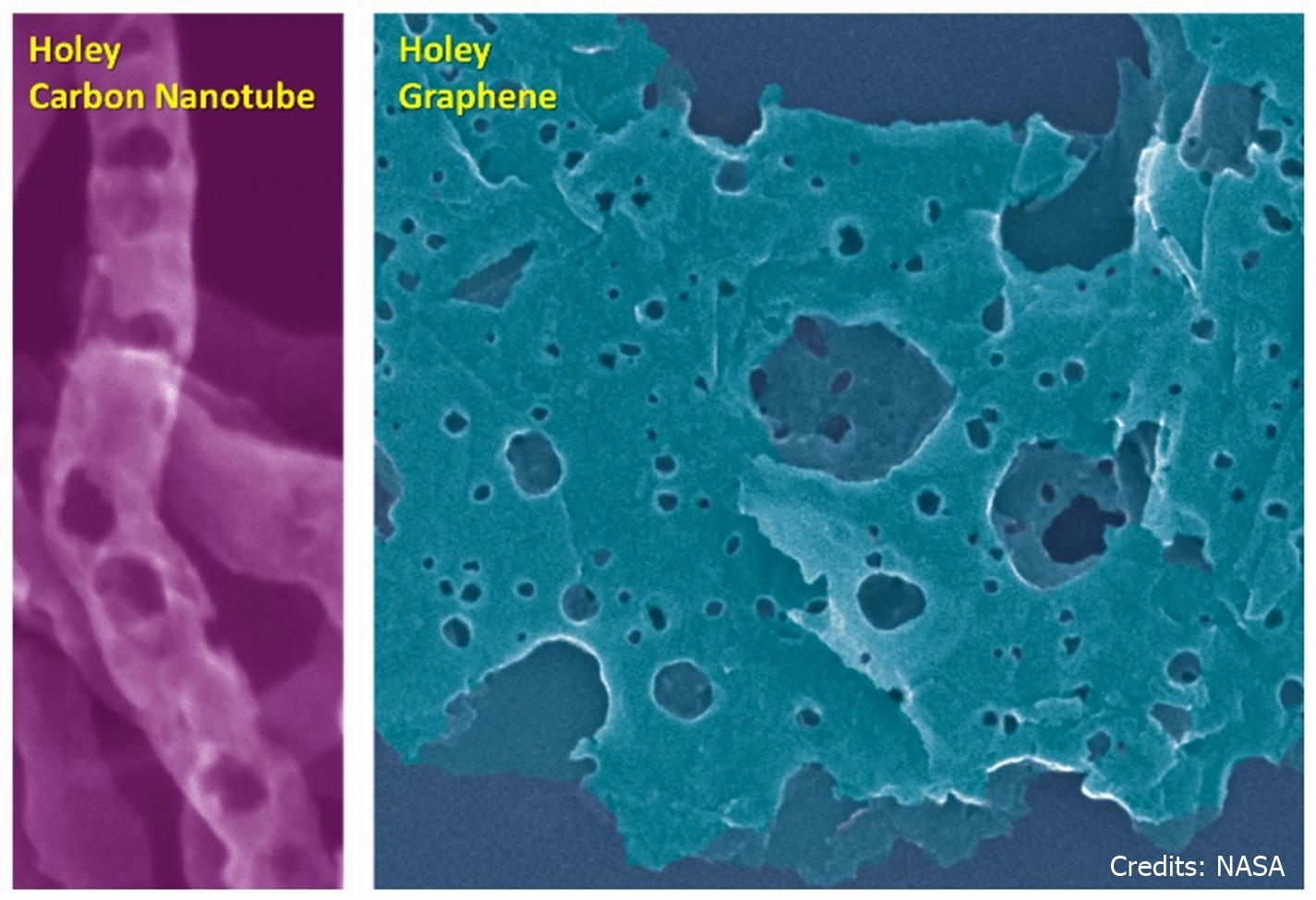
Benefits
- Produces carbon nanomaterials with increased surface area, improved electrochemical performance, and through-surface molecular transport properties
- Eliminates need for catalysts, solvents, and flammable gases for processing
- Consistently produces holes with a narrow size distribution, especially in the sub 10-nm ranges
- Allows for control of material properties such as surface area, modulus, thermal conductivity, and thermopower
- Is readily scalable and can be used to generate bulk quantities (only dependent on the availability/cost of pristine materials)
Applications
- Energy storage (supercapacitors, batteries)
- Membranes for gas separation, water desalination, biomolecular separation
- Gas and drug delivery
- Chemical and biological sensors
- Thermoelectrics
- Coatings
|
Tags:
|
|
|
Related Links:
|
Similar Results

Carbon Bipolar Membranes for Solid-State Batteries
In traditional batteries with liquid electrolytes, e.g., lithium-ion, each battery cell must be individually sealed, packaged, and electrically connected to other cells in the pack. The cells in solid-state batteries on the other hand may be stacked on top of one another with only a separation layer in between, called a bipolar plate. These bipolar plates or membranes if thin enough must be electrochemically inert to the electrode and electrolyte materials while providing electrical connectivity between the individual cells.
Here, NASA has combined advances in the preparation of carbon nanomaterials and solid-state batteries to create extremely lightweight bipolar plates and membranes. These bipolar membranes will enable high energy density solid-state batteries unachievable with typical bipolar plate materials like stainless steel, aluminum, aluminum-copper, or conductive ceramics. The carbon bipolar membranes may be fabricated in multiple ways including but not limited to directly compressing carbon powders onto an electrode-electrolyte stack or separately making a film of the carbon material and dry pressing the film between other battery layers. The new bipolar membranes have been demonstrated in high energy density solid-state batteries in coin and pouch cells.
The carbon bipolar membranes are at technology readiness level TRL-4 (Component and or breadboard validation in laboratory environment)and are available for patent licensing.

Metal Oxide-Vertical Graphene Hybrid Supercapacitors
The electrodes are soaked in electrolyte, separated by a separator membrane and packaged into a cell assembly to form an electrochemical double layer supercapacitor. Its capacitance can be enhanced by a redox capacitance contribution through additional metal oxide to the porous structure of vertical graphene or coating the vertical graphene with an electrically conducting polymer. Vertical graphene offers high surface area and porosity and does not necessarily have to be grown in a single layer and can consist of two to ten layers. A variety of collector metals can be used, such as silicon, nickel, titanium, copper, germanium, tungsten, tantalum, molybdenum, & stainless steel.
Supercapacitors are superior to batteries in that they can provide high power density (in units of kw/kg) and the ability to charge and discharge in a matter of seconds. Aside from its excellent power density, a supercapacitor also has a longer life cycle and can undergo many more charging sequences in its lifespan than batteries. This long life cycle means that supercapacitors last for longer periods of times, which alleviates environmental concerns associated with the disposal of batteries.

Holey Graphene Mesh from Solvent-Free Manufacturing and Composites Thereof
The HGM or composite HGM developed is a novel nanocarbon-based architecture that (1) is prepared from dry processing from commercially available starting materials or readily prepared composites thereof; (2) exhibits micropores and mesopores due to the holey graphene sheets and their stacking; (3) exhibits micron- and macro-sized pores in the article. The method can produce a range of high-fidelity hole size, shape, and distribution on the graphene or composite articles. The disclosed laser-based method is easily scaled-up and automatable. The result is a novel ultra-lightweight graphene-based mesh structure with high electrical conductivity, thermal conductivity, high surface area, high through-thickness unimpeded ion transport, mechanical robustness. The HGM-based composites utilize HGM as a novel framework, matrix, or substrate for secondary components that are active for energy storage, catalysis, sensing, optical, filtration, and biological applications.

Advanced Protective Coatings for Graphite-Based Nuclear Propulsion Fuel Elements
To protect the graphite (Gr)-based substrates in a nuclear systems fuel elements from hot hydrogen attacks, earlier researchers developed a method to deposit (via chemical vapor deposition) a protective niobium carbide (NbC) or zirconium carbide (ZrC) coating in the inner cooling channels of the fuel elements through which the hydrogen propellant flows. Unfortunately, the significant difference in the coefficients of thermal expansion (CTE) between the Gr-based substrate and the ZrC coating leads to debonding at intermediate temperatures, thereby exposing the substrate to hot hydrogen attack despite the NbC or ZrC coating. Innovators at Glenn have proposed a solution to this problem by introducing additional layers of compliant metallic coatings to accommodate the differences in CTE between the ZrC and the Gr-substrate,thereby potentially increasing life and durability. In this configuration,the innermost layer is composed of molybdenum carbide (Mo2C), and additional outer layers are made of molybdenum (Mo) and niobium (Nb) layers. The MoC acts as a diffusion barrier to minimize the diffusion of carbon into the refractory metal layers and the diffusion of Mo or Nb into the Gr-based substrate. The Mo layer is deposited on top of the Mo2C layer. A Nb layer is deposited on the Mo layer with the ZrC forming the outside layer of the coating. A thin Mo layer on the ZrC helps to seal the cracks on the ZrC and acts a diffusion barrier to hydrogen diffusion into the coating.The Mo and Nb layers are compliant so that differences in the thermal expansion of ZrC and other layers can be accommodated without significant debonding or cracking. They also act as additional diffusion barriers to hydrogen diffusion towards the Gr-substrate. Overall, Glenn's pioneering use of layered coatings for these components will potentially increase the durability and performance of nuclear propulsion rockets.

Cryogenic Flux Capacitor
Storage and transfer of fluid commodities such as oxygen, hydrogen, natural gas, nitrogen, argon, etc. is an absolute necessity in virtually every industry on Earth. These fluids are typically contained in one of two ways; as low pressure, cryogenic liquids, or as a high pressure gases. Energy storage is not useful unless the energy can be practically obtained ("un-stored") as needed. Here the goal is to store as many fluid molecules as possible in the smallest, lightest weight volume possible; and to supply ("un-store") those molecules on demand as needed in the end-use application. The CFC concept addresses this dual storage/usage problem with an elegant charging/discharging design approach.
The CFC's packaging is ingeniously designed, tightly packing aerogel composite materials within a container allows for a greater amount of storage media to be packed densely and strategically. An integrated conductive membrane also acts as a highly effective heat exchanger that easily distributes heat through the entire container to discharge the CFC quickly, it can also be interfaced to a cooling source for convenient system charging; this feature also allows the fluid to easily saturate the container for fast charging. Additionally, the unit can be charged either with cryogenic liquid or from an ambient temperature gas supply, depending on the desired manner of refrigeration. Finally, the heater integration system offers two promising methods, both of which have been fabricated and tested, to evenly distribute heat throughout the entire core, both axially and radially.
NASA engineers also applied the CFC to a Cryogenic Oxygen Storage Module to store oxygen in solid-state form and deliver it as a gas to an end-use environmental control and/or life support system. The Module can scrub out nuisance or containment gases such as carbon dioxide and/or water vapor in conjunction with supplying oxygen, forming a synergistic system when used in a closed-loop application. The combination of these capabilities to work simultaneously may allow for reduced system volume, mass, complexity, and cost of a breathing device.



